Depriester chart isobutane software#
Use Aspen Plus Software to Simulate Ex 1. function RRE, this is ABOVE the dew point… In this case, we will have something between 0 1 reduce T (which is a dew point temperature or pressure, respectively) (which is a bubble point temperature or pressure, respectively) If the specifications that you are given for your single-stage equilibrium separations For multicomponent, we are interested on calculating dew and bubble points Use the animation to verify certain conditions. There is only a SINGLE line that describes these characteristic

As you will see, if T/P are fixed, then, for pure substances: X values vary from substance to substance and the units being usedĬontact me if can also check out more content here: Note that each plot/graph of each hydrocarbons can be written in the form of The temperature and pressure of the system must specified Graph (called the “K chart”) prepared by DePriester For light hydrocarbons, the value of Ki of each species can be obtained from the

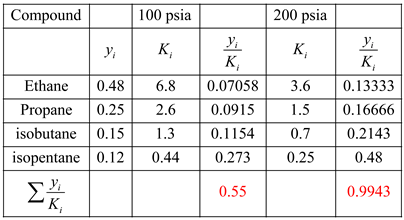
Verify K-Values of several Hydrocarbons It will now be convenient for us to work with K-Values in multicomponent systems High boiling point HC have LOW K-values Low boiling point HC have HIGH K-values The larger (heavier) the HC, the greater its BP, i.e. They will have mostly van der waal forces, i.e. According to chemistry, the hydrocarbons’ boiling point depends on their size, as Recall that K-Value is a relationship between liquid and vapor phases: In this section, we will cover only multiple-alkane systems For a Ternary (3 species in equilibrium) System, then we get: We have been studying binary systems, that is two species K-Values for Hydrocarbon Systems (dePriester) There I designed and modeled several processes relating separation of isopentane/pentane mixtures, catalytic reactors and separation processes such as distillation columns, flash separation devices and transportation of tank-trucks of product.įlash Distillation in Chemical and Process Engineering (Part 3 of 3) I worked as a Process Design/Operation Engineer in INEOS Koln, mostly on the petrochemical area relating to naphtha treating. I majored in Chemical Engineering with a minor in Industrial Engineering back in 2012. You will be able to continue with Batch Distillation, Fractional Distillation, Continuous Distillation and further courses such as Multi-Component Distillation, Reactive Distillation and Azeotropic Distillation. You will be able to understand mass transfer mechanism and processes behind Flash Distillation.
Material and Energy Balances for flash systemsĪnimations and Software Simulation for Flash Distillation Systems (ASPEN PLUS/HYSYS)Īll theory is taught and backed with exercises, solved problems, and proposed problems for homework/individual study. REVIEW: Of Mass Transfer Basics (Equilibrium VLE Diagrams, Volatility, Raoult's Law, Azeotropes, etc.)ĭistillation Theory - Concepts and PrinciplesĪpplication of Distillation in the IndustryĮquipment for Flashing Systems such as Flash Drums Understanding the concept behind Gas-Gas, Liquid-Liquid and the Gas-Liquid mass transfer interaction will allow you to understand and model Distillation Columns, Flashes, Batch Distillator, Tray Columns and Packed column, etc. Other Equilibrium Diagrams: P = 1.Binary Distillation is one of the most important Mass Transfer Operations used extensively in the Chemical industry. Phase Equilibrium (Alternate Form VLE) Historically, when estimates were done by hand: Sometimes the K values are nearly composition independent “hand” techniques of design/solution have used DePriester Charts (hydrocarbons):ĭePriester Chart P = 2 bar T = 100 oC Isobutane others…. Phase Equilibrium (Alternate Form VLE) Historically, when estimates were done by hand:
